Syfovre™ is a novel treatment for an advanced stage macular degeneration called geographic atrophy.
The FDA has approved Syfovre™ (pegcetocplan) for the treatment of GA, a leading cause of in developed countries. Syfovre™ is the first treatment for this advanced stage of macular degeneration. Treatment for wet macular degeneration has been available for over a decade.
Administration of Syfovre™
As with many retinal therpies, Syfovre™ is administered as an intravitreal injection into the eye. Because eye drops, ointments, oral meds and intravenous medications have difficulty penetrating the eye, intravitreal injections are necessary to deliver the right amount of drug to the retina.
Injections may be given monthly or every other month and will need to be repeated. The exact frequency of dosing will be determined by your doctor.
How Does Syfovre™ Work?
Pegcetocoplan is a unique inhibitor of C3, a protein involved in the complement system. The complement system is a major factor in our immune system and helps prevent infection from bacteria, viruses, regulates inflammation and aids the body get rid other pathogens.
Left unchecked, C3 may be involved in the death of healthy cells of the retina called retinal pigment epithelial (RPE) cells. RPE cells are responsible for the health and maintenance of photoreceptors, such as rods and cones. Without healthy photoreceptors, we can not see.
Syfovre™ binds C3 much like an antibody and prevents C3 from causing cell death to the retinal RPE cells.
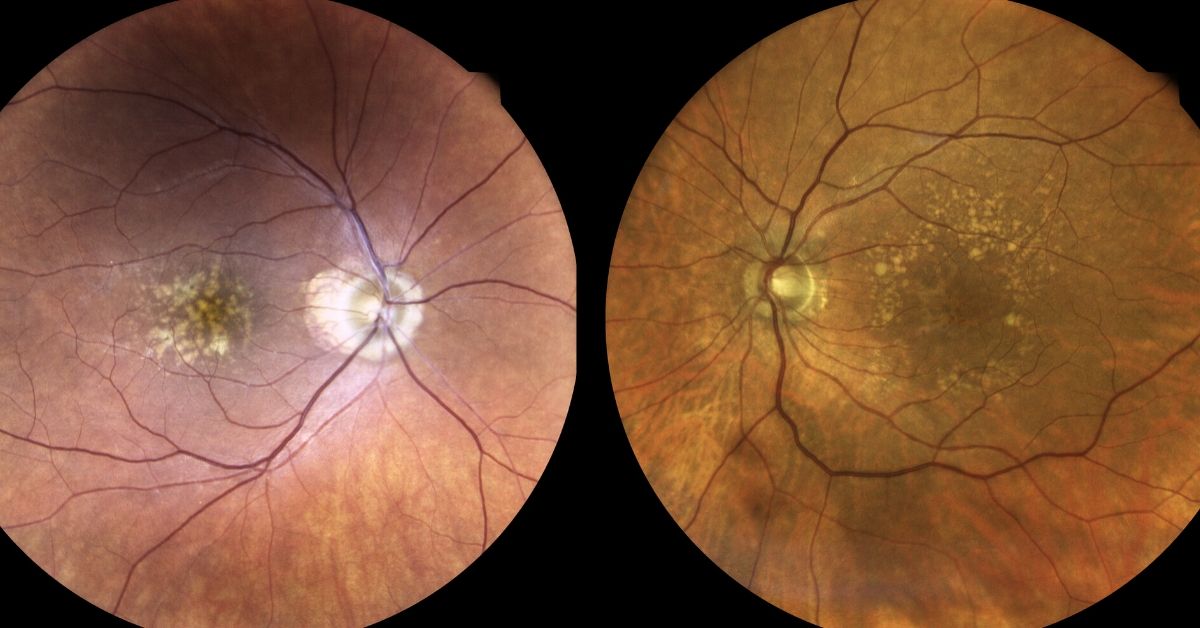
Geographic Atrophy
Areas of RPE cell loss can be detected during a retinal examination. Usually these so-called “atrophic lesions” arise near the macula. With time the lesions enlarge to involve the central macula.
Visual acuity, your central vision, is usually spared early in the disease, but visual function may be compromised depending upon the size, number and location with respect to the central macula.
Unlike wet macular degeneration where abnormal blood vessels develop within the layers of the retina, geographic atrophy is causes by death of the RPE cells.
Symptoms of Geographic Atrophy
Symptoms of macular degeneration include: blurry vision, distortion and loss of central vision. Geographic atrophy can also cause decline in visual function such as poor contrast, decreased reading speed, poor color perception etc.
If you or your family member is concerned, please call your eye care provider. The earlier this is detected and possibly treated, the better.
If you would like to schedule an appointment, please call us (877) 245.2020.